![]()
Article
Synthesis of silver nanoparticles from Sargassum tenerrimum and screening phytochemicals for its anti-bacterial activity
P.Kumar 1, S.Senthamil Selvi 2, A.Lakshmi Prabha 2, K.Prem Kumar 3, R.S.Ganeshkumar 1 and M. Govindaraju 1*
1 Department of Environmental Biotechnology, Bharathidasan University, Tiruchirappalli – 24, India.
2 Department of Plant Science, Bharathidasan University, Tiruchirappalli, India.
3 Department of Biomedical Science, Bharathidasan University, Tiruchirappalli, India.
* Corresponding author. Email: mgrasu@gmail.com
Citation: P. Kumar, et al. Synthesis of silver nanoparticles from Sargassum tenerrimum and screening phytochemicals for its anti-bacterial activity. Nano Biomed. Eng. 2012, 4(1), 12-16.
DOI: 10.5101/nbe.v4i1.p12-16.
Abstract
Synthesis of nanomaterials by biological approach is innovative, cheaper and environmental friendly and requires less-labor. In this regard, the present study focused on the synthesis of silver nanoparticles from the extract of Sargassum tenerrimum. Synthesized silver nanoparticles were well-characterized by UV-Visible Spectroscopy (UV-Vis), Fourier-Transform Infra-red Spectroscopy (FT-IR), Transmission Electron Microscopy analysis (TEM) and Dynamic Light Scattering (DLS). It was found that spherical shaped nanoparticles of size 20 nm were found in TEM analysis and showed effective anti-bacterial activity against standard reference strains. Altogether, extracts from seaweed were screened for phytochemicals followed by FT-IR prediction to reveal chemical functional groups present. The results showed that the anti-bacterial activity of silver nanoparticles was comparably higher than the phytochemicals present. Therefore, the present study elucidates silver nanoparticles can play a vital role in nano-based therapy in future.
Keywords: Sargassum tenerrimum, Silver nanoparticles, Phytochemicals, TEM, Anti-bacterial activity
1. Introduction
The non-polluting nanotechnologies have revolutionized the production of nanomaterials as environmentally safe products. Several chemicals used in the synthesis of nanoparticles are toxic which leads to environmental pollution [1]. Therefore, an alternative methodology is mandatory to trounce the toxic and polluting chemicals, along with various reducing and stabilizing agents. The naturally available biological resources can be an alternative source for the biosynthesis of nanoparticles [2, 3, 4]. Seaweeds are group of marine plants that are rich source of biologically active substances with biomedical applications. Hence, biosynthesis of nanoparticles using seaweeds has turned much of our attention towards the utilization of renewable marine resources. Historically, silver metal has been widely used as a therapeutic agent for curing diseases. In addition, soluble silver salts are more advantageous over epilepsy, nicotine addiction, gastroenteritis, stomatitis [5, 6] and sexually transmitted diseases [7]. Consequently, biosynthesized silver nanoparticles have a promising role in biomedical applications. It is well understood that silver nanoparticles possess elevated surface area that hinder microbial growth compared to silver metal as a bulk [8]. These nanoparticles can directly interact with bacterial cells causing oxidative stress that result in deadly damage [9]. The anti-bacterial activity of silver nanoparticles depends upon the bioavailability of silver ions [10] and due to the presence of reducing agents [2]. With reference to the biological perspectives, we report on the biosynthesis of silver nanoparticles from marine brown seaweed, Sargassum tenerrimum. Recently, polysaccharides from Sargassum tenerrimum showed anti-viral activity against Herpes Simplex Viruses (HSVs) [11]. In addition, anti-bacterial potential of brown seaweeds is impressive over human pathogens [12]. Researchers predict that bio-reduction of metal compounds by plants is due to the presence of phytochemicals [2]. Water-soluble phytochemicals such as polyphenols, flavones, organic acids and quinones are greatly responsible for immediate reduction. Taking this as an initiative, we aimed to instigate the anti-bacterial efficacy of rapidly biosynthesized silver nanoparticles from Sargassum tenerrimum with that of its phytochemical constituents.
2. Materials and Methods
2.1 Preparation of aqueous seaweed extract
Sargassum tenerrimum was collected by hand picking method at a depth of 1–2 meter in Gulf of Mannar, Mandapam (9°16’47”N, 79°7’12”E), India. The seaweed was surface sterilized with tap water to remove extraneous substances followed by distilled water. The seaweed is identified, shade dried for 15 days and powdered using mixer grinder. Later, aqueous extract was prepared by dissolving 200 mg of powdered seaweed in 100 ml of sterile distilled water. The extract was heated at 60°C for 20 mins, centrifuged at 8,000 rpm and the supernatant was used throughout the study.
2.2 Synthesis and characterization of silver nanoparticles (AgNPs)
In an Erlenmeyer’s flask, 95 ml of 1 mM Silver nitrate (AgNO3) solution was prepared using deionized water and 5 ml of aqueous seaweed extract was added [13]. The reaction mixture was gradually heated to 90°C for 20 mins and the change in color was envisaged as the evidence of bioreduction. Characterization of silver nanoparticles was performed in sequence using UV-Visible Spectrophotometer (Shimadzu - 2450), FT-IR (Spectrum RX-1 instrument) and Transmission Electron Microscopic (Tecnai 10 instrument). The particle size distribution and zeta potential analysis of silver nanoparticles was evaluated using Dynamic Light Scattering measurement (Malvern – zetasizer – nano-Z590).
2.3 Preliminary screening of phytochemicals
Powdered seaweed of 10 g was soaked in 100 ml of methanol to obtain crude extract. The mixture was then centrifuged at 5000 rpm for 20 minutes at 4°C and filter sterilized using 0.2 µm membrane syringe. A fraction of seaweed extract was subjected to phytochemical screening as described by Harborne, 1998 [14]. Natural chemical groups such as amino acids, alkaloids, carbohydrates, flavonoids, saponins, sterols, tannins, terpenoids, proteins and phenolic compounds were probed. The methanolic extract was further exemplified by FT-IR spectroscopic studies to reveal the characteristic functional group present.
2.4 Anti-bacterial efficacy by disk diffusion method
Bacterial cultures such as Bacillus cereus (MTCC 1272), Escherichia coli (MTCC 1687), Klebsiella pneumoniae (MTCC 530), Proteus mirabilis (MTCC 425), Pseudomonas aeruginosa (MTCC 1688), Salmonella typhii (MTCC 531), Shigella flexneri (MTCC 1457) Staphylococcus aureus (MTCC 96) and Vibrio cholerae (MTCC 3906) were obtained from Microbial Type Culture Collection (MTCC), Indian Institute of Microbial Technology, Chandigarh, India. Disk diffusion method was adopted to evaluate the anti-bacterial efficacy of aqueous extract, biosynthesized silver nanoparticles and methanol extract from the Sargassum tenerrimum along with 1 mM of silver nitrate as positive control. Freshly prepared Luria Bertani (LB) agar plates were inoculated with exponential bacterial cultures (106 CFU/ml) by spread plate technique. Sterile antibiotic disks (HIMEDIA – 6 mm) were impregnated with 30 µl of test samples and placed on the agar plates. The plates were incubated at 37°C for 24 h and Zone of Inhibition (ZoI) was measured, mean while the experiments were performed in triplicates.
3. Results and Discussion
3.1 Synthesis and characterization of silver nanoparticles
Several reports have been employed for the synthesis of silver nanoparticles for its beneficial applications. Recently, seaweeds have been identified as the potential source for synthesizing nanoparticles while Singaravelu et al [3] have synthesized gold nanoparticles from Sargassum wightii extract within 24 h of incubation time. Similarly, we have synthesized silver nanoparticles from the extract of Sargassum tenerrimum rapidly within 20 min. Endpoint with prominent color change (Fig. 1) indicates the excitation of surface plasmon resonance due to reduction of silver nitrate [14]. Longitudinal plasmon vibrations corresponding to silver nanoparticles were convincing with UV spectral peak at 420 nm and with no absorption peak in control i.e. without reductant (Fig. 2). This clearly indicates the interaction between silver ions and biomolecules present in the aqueous seaweed extract. Intensity of band increased upon varying time without any shift in peak position. According to transmission electron microscope, the morphology of silver nanoparticles observed to be spherical with an average size of 20 nm (Fig. 3). Particle size distribution was plotted using the results obtained from Dynamic Light Scattering analysis (DLS) with a maximum intensity at 45 nm (Fig. 4a) and zeta potential measurements disclosed that the silver nanoparticles are highly stable with a value of –27mV (Fig. 4b). FT-IR bands corresponding to silver nanoparticles are at 3431 cm-1, 2386 cm-1, 2073 cm-1, 1637 cm-1 and 693 cm-1 (Fig. 5a). The absorption band corresponding to 3431 cm-1 is due to N-H stretching vibrations of peptide linkages and hydroxyl stretch vibrations of carboxylic acid groups demonstrating the presence of polyphenols [15]. Further, amide - I band which is due to carbonyl stretch vibrations in the amide linkages of protein at 1637 cm-1 representing polyphenols [16]. The CO peak at 2386 cm-1 and 2073 cm-1 decreases its intensity and a peak formed at 692 cm-1 is may be attributed to out of sketch blending of aromatic ring

Fig. 1. Silver nanoparticles using Sargassum tenerrimum a) before synthesis (control), b) after synthesis (synthesized silver nanoparticles)
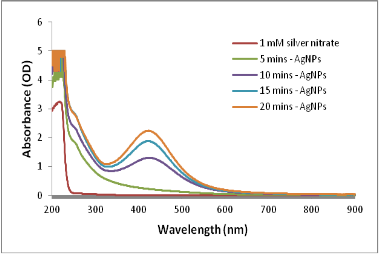
Fig. 2. UV-Vis-spectra of silver nanoparticles.

Fig. 3. TEM images obtained after 10 fold dilution of purified silver nanoparticles using Sargassum tenerrimum. The range of observed diameter was 100 nm.
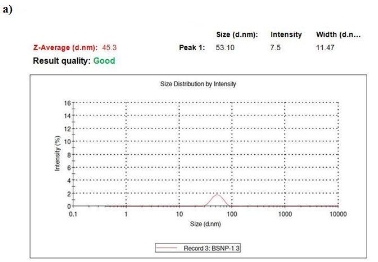
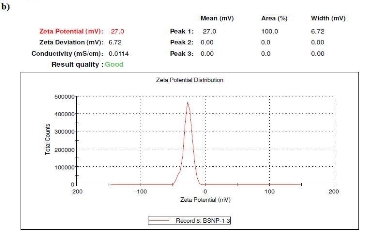
Fig. 4. Dynamic Light Scattering measurements a) Size distribution by intensity, b) Zeta potential measurements.
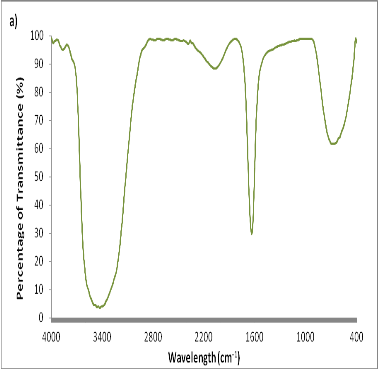
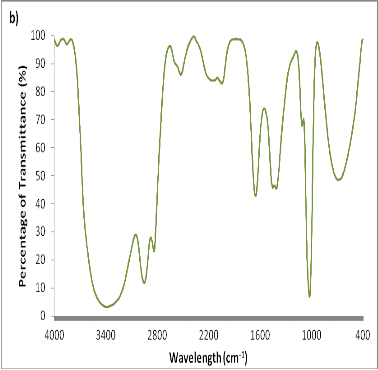
Fig. 5. FT-IR spectral patterns of Sargassum tenerrimum ranging from 4000-400 cm-1. a) Silver nanoparticles b) Phytoconstituents.
Table 1. Preliminary Screening of Phytochemicals
(+) presence (-) absence
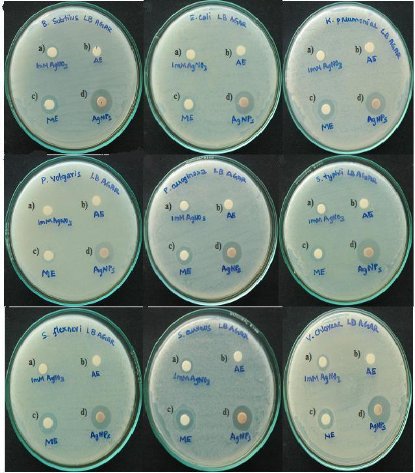
Fig. 6. Anti-bacterial activity of various extracts against nine human pathogenic strains. a) 1 mM silver nitrate (1mM AgNO3 - positive control) b) Aqueous Extract (AE) c) Methanol Extract (ME) d) Biosynthesized silver nanoparticles (AgNPs)
3.2. Phytochemical analysis
The preliminary phytochemical analysis of methanol extract revealed the presence of amino acids, alkaloids, carbohydrates, flavonoids, saponins, sterols, tannins, terpenoids, proteins, and phenolic compounds as shown in Table. 1. FT-IR predicts the molecular configuration of different functional group present in the seaweed extract. Considerable absorption peak were found at 3387 cm-1, 2951 cm-1, 2838 cm-1, 2524 cm-1, 2155 cm-1, 2044 cm-1, 1652 cm-1, 1452 cm-1, 1412 cm-1, 1110 cm-1, 1023 cm-1 and 686 cm-1 respectively (Fig. 5b). The peak corresponding to 3387 cm-1 indicates the presence of intermolecular hydrogen bonding with hydroxyl group with polymeric association. The peak formed at 2951 cm-1, 2838 cm-1 is because of C-H stretching and symmetric stretching of methoxy groups present [17]. A small peak at 2524 cm-1 is an attribute to S-H stretching vibration mode. The stretch between 2155 cm-1 – 2161 cm-1 typically formed by triple C ≡ C bond in alkynes or because of cumulative double bond in ketones [18]. A minute peak with less intensity at 1451 cm-1 indicates the presence of asymmetric CH3 bending modes of the methyl groups of proteins [19]. The absorption peak formed at 1412 cm-1 is due stretching C-N, deformation N-H and deformation of C-H [16]. Peak at 1110 cm-1 and 1024 cm-1 is responsible for polysaccharides [20] and C-O stretch associated with glycogen [21] respectively.
3.3 Comparative anti-bacterial activity
Unlike previous reports [2, 3, 4], comparative anti-bacterial property of silver nanoparticles and methanol extract was executed. The test was carried out against nine pathogenic reference strains including both Gram-positive and Gram-negative bacteria (Fig. 6). Silver nanoparticles exhibited good anti-bacterial activity (more than 15 mm) against all pathogens specifically to Vibrio cholerae (18 mm) which is more susceptible (Fig. 7). The aqueous extract of seaweed was found to have unclear zone of inhibition compared to 1mM silver nitrate as positive control. This illustrates that, silver compounds are toxic to microorganisms with strong anti-bacterial effect including multi-drug resistant bacteria [22]. Similarly, electrostatic interaction between positively charged nanoparticles and the negatively charged bacterial membrane induces cell permeability resulting in cell death [23]. Besides, methanol extract proves its efficiency when compared to aqueous extract due to the presence of secondary metabolites. Rao et al [24] have reported that 12 different species of Sargassum extract possess high anti-bacterial activity against human pathogenic bacteria. Similarly, Shelat [25] have found that methanol extract of Sargassum plagiophyllum was found to be active against Gram-positive bacteria while acetone extract of Sargassum tenerrimum showed high activity against all tested strains. The interesting information is that, green synthesis of silver nanoparticles by exploiting seaweed has immense anti-bacterial activity compared to all other extracts. The study also suggests that seaweeds containing phytochemicals can be better opted for nanoparticle synthesis. This work throws some light on futuristic research work on synthesis of nanoparticle from seaweeds towards biomedical applications.
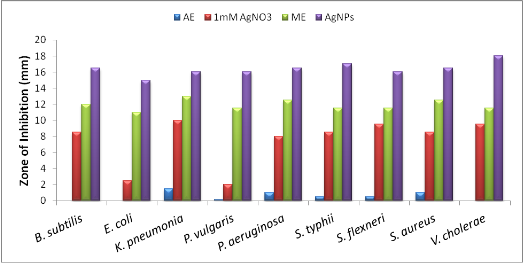
Fig. 7. Comparative anti-bacterial activity based on Zone of Inhibition (mm). a) Aqueous Extract (AE), b) 1 mM silver nitrate (AgNO3 – positive control), c) Methanol Extract (ME), d) Biosynthesized silver nanoparticles (AgNPs)
4. Conclusions
To summarize, comparative anti-bacterial activity of silver nanoparticles and phytochemicals from Sargassum tenerrimum was evaluated. Silver nanoparticle were synthesized and characterized in ambient conditions with an average size of 11–20 nm. The presence of phytochemicals as reducing agents in synthesizing nanoparticle can be potent antimicrobials in near future. The result concluded that biosynthesized nanoparticles showed evidence of high anti-bacterial activity against all test pathogenic strains compared to phytochemical constituents. A further study is needed to find out the actual inhibitory mechanism of silver nanoparticles.
References
Copyright: (c) 2012 P. Kumar, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.