Research Article
Epoxidized Palm Oil Plasticized Polycaprolactone Nanocomposites Preparation
Emad Abbas Jaffar Al-Mulla 1*, Fayq Hsan Jabbar 2, Zaidoon Jawad Kadhim 3, Ali Abdulabbas Abdullah 4, Ahmed Ghanim Wadday 4, Sabah Mohammed Mlkat 5
1 Department Pathological Analysis, College of Health and Medical Techniques, Al-Furat Al-Awsat Technical University, 54003 Al-Kufa, Iraq.
2 Department of Research and Development, Alkarkh University of Science, Baghdad, Iraq.
3 Department of Chemistry, College of Science, University of Missan, Iraq.
4 Engineering Technical Faculty, Al-Furat Al-Awsat Technical University, An-Najaf, Iraq.
5 Electrical Department, Al-Furat Al-Awsat University, Technical Institute of Samawa, Samawa, Iraq.
* Corresponding author. E-mail: almullaemad@gmail.com
Received: Aug. 15, 2017; Accepted: Sep. 14, 2017; Published: Sep. 20, 2017
Citation: Emad Abbas Jaffar Al-Mulla, Fayq Hsan Jabbar, Zaidoon Jawad Kadhim, Ali Abdulabbas Abdullah, Ahmed Ghanim Wadday, and Sabah Mohammed Mlkat, Epoxidized Palm Oil Plasticized Polycaprolactone Nanocomposites Preparation. Nano Biomed. Eng., 2017, 9(3): 214-220.
DOI: 10.5101/nbe.v9i3.p214-220.
Abstract
As alternatives to petroleum-based polymeric materials, biodegradable polymers, such as polycaprolactone has attracted a lot of attention in the scientific community due to a rapid growth of intensive interest in the global environment. Chalcone, hexadecylamine and tetradecylamine were used as one of the organic compounds to modify natural montmorillonite clay. The clay modification was carried out by stirring the clay particles in an aqueous solution of chalcone-montmorillonite, hexadecylamine-montmorillonite and tetradecylamine-montmorillonite increasing from 1.29 to 1.53, 1.59 and 1.79 nm, respectively. The modified clay was then used in the preparation of the polycaprolactone/epoxidized palm oil blend nanocomposites. They were prepared by incorporating 0.5 - 5% of chalcone-montmorillonite, hexadecylamine-montmorillonite and tetradecylamine-montmorillonite. The interaction of the modifier in the clay layer was characterized by X-ray diffraction and transmission electron microscopy. The nanocomposites were synthesized by solution casting of the modified clay and a polycaprolactone/epoxidized palm oil blend at the weight ratio of 80/20, which had the highest increase in the tensile strength of the blend. The X-ray diffraction and transmission electron microscopy results confirmed the production of nanocomposites. The results also showed higher thermal stability for nanocomposites compared to those of the polycaprolactone /epoxidized palm oil blend.
Keywords: Nanocomposites; Surfactants; Organic cations; MMT; PCL; EPO
Introduction
The polycaprolactone (PCL) appears to be most attractive because of its availability and biodegradability [1, 2]. Many efforts have been made to discover, develop and modify biodegradable polymers derived from renewable resources [3]. The PCL is one of the biodegradable thermoplastic polyesters which can be prepared from butanediol and succinic acid produced by fermentation process [4, 5]. In addition to its applications in textile industry and medical fields, PCL is a promising candidate to produce disposable packaging. However, low molecular weight, low stiffness and high cost restrict its applications [6]. Many studies have been conducted to enhance the properties by blending biodegradable polymers with other polymers or using low molecular weight plasticizers [7–13]. The morphologies and phase behaviors of polylactic acid / PCL blend have been investigated [14]. The epoxidized palm oil (EPO) is used to improve properties and biodegradation of PCL. EPO is produced from esters of glycerol in palm oil containing different saturated and unsaturated fatty acids. It offers many advantages in chemical industries field because it is derived from renewable, biodegradable and abundant raw materials [9, 16-18]. The incorporation of organoclays in the polymer to produce a nanocomposite is another means to modify the property balance of a material. The improvements in thermal stability, physical and mechanical properties can be achieved by addition of 0.5 - 5% weight of organoclays in comparison to the neat polymer [19, 20]. The montmorillonite (MMT) modification may carry out by exchanging the original interlayer cations of organic cations where they are transformed from organophobic to organophilic materials and significantly increase the basal spacing of the clay layers [21]. It is generally accepted that the extent of swelling depends on the length of the alkyl chain and the cationexchange capacity of the clay [22]. Organoclays are mainly obtained by exchanging cations in the clay minerals, which contain hydrated Na+ ions with alkylammonium [23, 24]. It is reported that the organo-modified MMT-based composites have shown the possible competition between the polymer matrix and the plasticizer for the intercalation between the aluminosilicate layers [25]. In the present study, chalcone (CH), hexadecylamine (HAD) and tetradecylamine (TDA), as ammonium compounds, were used to modify the compatibility of MMT clay with EPO plasticized polycaprolactone to produce nanocomposites.
Experimental
Materials
MMT, HAD and TDA were obtained from Sigma Aldrich, Germany. EPO was provided by Advanced Oleochemical Technology Division (AOTD), Malaysia. PCL and chloroform were purchased through local suppliers from Nagoya, Japan and Merck, Germany, respectively. P-aminoacetophenone and p-chlorobenzaldehyde were obtained from Fluka, Germany. Sodium hydroxide and hydrochloric acid were obtain from J.T. Baker, USA.
Methods
Preparation of Chalcone (CH) was carried out according to procedure described by Sadiq et al., 2015 [26].
Preparation of organoclay
Organoclay was prepared with a cationic exchange process, where Na+ in the MMT was exchanged with alkylammonium ion from CH, HDA and TDA, which was reported in our previous paper [27], in an aqueous solution. 4.00 g of MMT was stirred vigorously in 600 mL of hot distilled water for 1 h to form a clay suspension. Subsequently, a designated amount of CH, HDA and TDA compounds, which had been dissolved in 400 mL of hot water and the desired amount of concentrated hydrochloric acid (HCl) was added into the clay suspension of CH, HDA and TDA compounds. After being stirred vigorously for 1 h at 80 ̊C, the organoclay suspension was filtered and washed with distilled water until no chloride was detected with 1 mol silver nitrate solution. It was then dried at 60 ̊C for 72 h. The dried organoclay was ground until the particle size was less than 100 μm before the preparation of the nanocomposite. Scheme1 is the schematic drawing of the nanocomposites production.
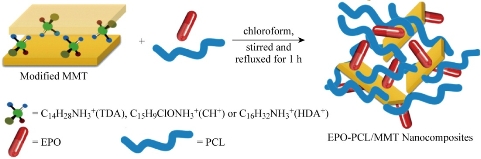
Scheme 1 Schematic drawing of EPO-PCL/MMT nanocomposites.
Preparation of PCL/EPO-clay nanocomposites
The required amounts of PCL and EPO were dissolved in 50 mL of chloroform, separately. The EPO solution was then transferred into the PCL solution with a dropper and continuous stirring. After all the EPO solution was transferred into the PCL solution, the resultant mixture was then stirred for 1 h. The required modified clay (CH –MMT, HDA-MMT and TDA-MMT) was then added into the dissolved PCL/EPO in the small portion. The mixture was then refluxed for 1 h and then ultrasonically stirred using the ultra sonic cathode for 10 min to make sure that the clay was fully dispersed in the PCL/EPO solution. The nanocomposite was then poured into a Petri dish and left to dry. The amounts of PCL/EPO and the modified clay used in this study are listed in Table 1.
Table 1 Amounts of PCL/EPO and the modified clay
|
Sample |
Weight of PBS (g) |
Weight of EPO (g) |
Weight of organoclay (g) |
|
1 |
0.9 |
0.1 |
0.00 |
|
2 |
0.86 |
0.09 |
0.05 |
|
3 |
0.82 |
0.08 |
0.1 |
|
4 |
0.78 |
0.07 |
0.15 |
|
5 |
0.74 |
0.06 |
0.2 |
|
6 |
0.50 |
0.25 |
0.25 |
Characterization
Measurements of tensile properties
The tensile strength was measured by using Instron Universal Testing Machine 4301 at 5 mm/min of crosshead speed, in accordance with ASTM D638 [2]. Seven samples were used for the tensile test and an average of five results was taken as the resultant value.
X-ray diffraction (XRD) analysis
X-ray diffraction (XRD) study was carried out using Shimadzu XRD 6000 diffractometer with Cu Kα radiation (0.15406 nm). The diffractogram was scanned in the ranges from 2◦ to 10◦ at a scan rate of 1◦/min.
Thermogravimetric analysis (TGA)
The thermal stability of the samples was studied by using Perkin Elmer TGA 7 Thermogravimetric Analyzer. The samples were heated from 35 to 500 ◦C with a heating rate of 10 ◦C/min under a nitrogen atmosphere with a nitrogen flow rate of 20 mL/min.
Transmission electron microscopy (TEM)
The dispersion of clay was studied by using energy filtering transmission electron microscopy (EFTEM). TEM pictures were taken in LEO 912 AB energy filtering transmission electron microscope with an acceleration voltage of 120 keV. The specimens were prepared using a Ultracut E (Reichert and Jung) cryomicrotome. Thin sections of about 100 nm were cut with a diamond knife at −120 ◦C.
Results and Discussion
Mechanical properties
The weight ratio of the PCL/EPO blend was fixed at 80 : 20 according to preliminary investigation which gave the highest tensile strength of the blend. Therefore, this ratio was chosen in subsequent experiments. Table 2 shows the effect of clay content on the tensile strength properties of the PCL/EPO/HAD-modified montmorillonite (OMMT), PCL/EPO/TAD-OMMT and PCL/EPO/CH-OMMT nanocomposite. The tensile strength of the PCL/EPO blend decreased to 11.89 MPa. The addition of 0.5, 1 or 2 php of OMMT into the PCL/EPO blend increased the tensile strength. The highest tensile strength (23,23 MPa) was observed with 2 php of HAD- OMMT; tensile strength 24,15 was observed with 2 php of TAD- OMMT; and tensile strength 21.71 was observed with 2 php of CH- OMMT.
Table 2 Tensile properties of PCL/EPO blend, PCL/EPO/HAD-OMMT, PCL/EPO/TAD-OMMT and PCL/EPO/CH-OMMT nanocomposite
|
Tensile strength (Mpa) |
Sample |
||
|
CH-MMT |
TAD-MMT |
HAD-MMT |
|
|
10.81 ± 0.14 |
13.29 ± 0.31 |
10.79 ± 0.29 |
PCL/EPO |
|
13.12 ± 0.32 |
14.02 ± 0.43 |
12.75 ± 0.31 |
PCL/EPO/0.5php OMMT |
|
16.05 ± 0.71 |
18.41 ± 0.72 |
17.01 ± 0.92 |
PCL/EPO/1.0php OMMT |
|
21.71± 0.72 |
24,15 ± 0.92 |
23.23 ± 1.25 |
PCL/EPO/2.0 php OMMT |
|
17.69 ± 0.61 |
18.25 ± 0.78 |
18.98 ± 1.10 |
PCL/EPO/3.0 php OMMT |
|
14.15 ± 0.49 |
14.05 ± 0.61 |
14.11 ± 0.80 |
PCL/EPO4.0 php OMMT |
|
11.61 ± 0.37 |
11.85 ± 0.56 |
10.92 ± 0.41 |
PCL/EPO/5.0 php OMMT |
Table 3 Diffraction angle and basal spacing of natural clay (MMT) and modified clays with the CH –MMT, HDA-MMT and TDA-MMT.
|
Sample |
Exchanged cation |
2ϴ (ͦ ) |
D(001) spacing (nm) |
|
MMT |
Na+ |
6.87 |
1.29 |
|
CH |
C15H9ClONH3+(CH+) |
5.83 |
1.53 |
|
HAD |
C16H32 NH3+ (HDA+) |
5.61 |
1.59 |
|
TDA |
C14 H28 NH3+(TDA) |
4.92 |
1.79 |
XRD analysis
According to Bragg’s law (n λ = d sin ϴ),where d refers to the distance of two consecutive clay layers, and λ is the wavelength of the intercept X-rays at the incident angle ϴ. The presence of CH-MMT, HDA-MMT and TDA-MMT chains in the galleries turned the original hydrophilic silicate to an organophilic silicate, and thus increased the layer-to-layer spacing of MMT [28, 29]. The interlayer distance of MMT was assigned with a basal spacing of 1.27 nm due to a d001 diffraction peak at 2ϴ = 6.91̊ resulted from X-ray diffraction. The CH, HDA and TDA were treated with MMT through a cation exchange process. The cationic head groups of the intercalation agent molecule would preferentially reside at the surface layer and the aliphatic tail would radiate a ways from the surface. Fig. 1 indicates that CH, HDA and TAD were successfully intercalated into the MMT galleries. The maximum basal spacing of CH-MMT, HDA-MMT and TDA-MMT increased from 1.29 to 1.53, 1.59 and 1.79 nm, respectively (Table 3). The XRD patterns of the nanocomposites prepared by using CH, HDA and TDA (alkylammonium compounds) modified MMT nanocomposites are shown in Fig. 2. In the nanocomposites, the basal spacing of the clay increased to 2.19, 2.59 and 2.79 nm for 80PCL20EPO/CH-MMT, 80PCL20EPO/HDA-MMT and 80PCL20EPO/TDA-MMT, respectively (Table 4). It was observed by Agag et al. that the basal spacing of organoclay in the polymer matrix increased with the increase in the size of the surfactant [30].
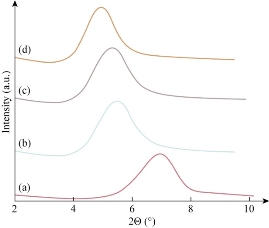
Fig. 1 The XRD patterns of (a) MMT, (b) CH-MMT, (c) HDA-MMT, and (d) TDA-MMT.
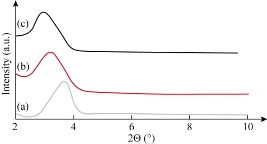
Fig. 2 XRD patterns of (a) PCL/EPO/CH, (b) HDA and (c) TDA modified clay nanocomposites.
Table 4 Diffraction angle and basal spacing of PLA/EPO/CH, HDA and TDA modified clay nanocomposites.
|
Sample |
2ϴ (ͦ ) |
d- spacing (nm) |
|
80PCL20EPO/CH-MMT |
4,12 |
2.19 |
|
80PCL20EPO/HDA-MMT |
3.41 |
2.59 |
|
80PCL20EPO/TDA-MMT |
3.34 |
2.79 |
TGA
Thermal degradation of MMT showed two steps [24]. The first one was before 200 ◦C because of the volatilization of water adsorbed on the external surfaces of the MMT and water inside the interlayer space. Decomposition of the surfactant took place in the second step between 200 and 500 ◦C. Fig. 3 illustrates the weight loss curves (TGA) of the MMT, CH-MMT, HDA-MMT and TDA-MMT in MMT containing water due to hydrated sodium (Na+) cations intercalated inside the clay layers. The presence of alkylammonium groups within the MMT interlayer spacing lowered the surface energy of the inorganic structure and would transform organophobic materials to organophilic ones. The major difference between the thermogram of the unmodified clay and that of the organoclay was that the organic constituents in the organoclay decomposed in the range from 200 to 500 ◦C, as the organic constituent in the organoclay decomposed in this range. The C decomposed as the temperature increased from 182 to 372 ̊C. The decomposition process ended at 372 ̊C. The CH started decomposing at higher temperatures than the TDA did, which started at 198 ̊C and ended at 349 ̊C. HDA had decomposition temperature (starting at 158 ̊C and ending at 308 ̊C). Higher decomposition temperatures of the CH-MMT, HDA-MMT and TDA-MMT were observed compared to neat MMT, which indicated that there was a strong intermolecular interaction between the alkylammonium cations and the clay. Thermal properties were tested on PCL/EPO/HDA-MMT nanocomposites in order to determine the effect of modified clay content in the polymer matrix. The onset of the degradation of the nanocomposites was higher, i.e. 258, 261, and 267 ̊C for PCL/EPO containing HDA-MMT, TDA-MMT and CH-MMT, respectively, compared to the PCL/EPO blend (249 ̊C) (Fig. 4). The thermal stability increased with the addition of the HDA-MMT, TDA-MMT and CH -MMT. The presence of silicate layers dispersed homogeneously in the polymer sheet, hindering the permeability of volatile degradation products out from the materials and helping delay the degradation of the nanocomposites.
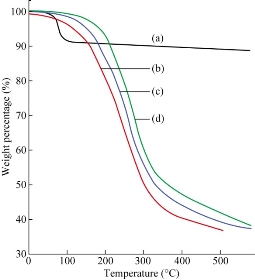
Fig. 3 TGA of (a) Na- MMT, (b) HDA-MMT, (c) TDA-MMT and (d) CH-MMT.
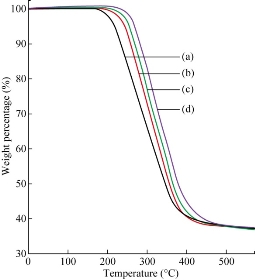
Fig. 4 TGA thermograms of (a) PCL/EPO blends, (b) PCL/EPO/HDA-MMT, (c) PCL/EPO/TDA-MMT and (d) PCL/EPO/CH-MMT
TEM
Nanocomposites based materials were further analyzed by TEM technique. PCL/EPO nanocomposites modified by employing CH, HDA and TDA showed a high level of intercalation and exfoliation of the silicate layers (Fig. 5). MMT stack morphology was fully preserved with PCL/EPO due to the incompatible nature of both constituents [27]. The dark lines represented the thickness of the individual clay layers or agglomerates.
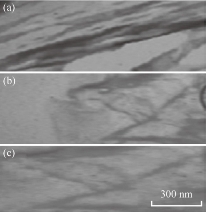
Fig. 5 TEM of (a) PCL/EPO/HDA-MMT, (b) PCL/EPO/TDA-MMT and (c) PCL/EPO/CH-MMT nanocomposites.
Conclusions
PCL/EPO modified MMT nanocomposites were prepared effectively by melt blending process. The silicate layers of the organoclay (2%) were intercalated in the polymer matrix using CH, HDA and TDA as modifiers for MMT. The formation of nanocomposites was confirmed by XRD and TEM techniques. In addition to their applications in textile industry and medical fields, biopolymer nanocomposites prepared in this study are a promising candidate to produce disposable packaging due to their good thermal properties and processability, and potentially to be produced from renewable, biodegradable, environmental friendly and easily available raw materials.
Conflict of Interests
The authors declare that no competing interest exists.
References
Copyright© 2017 Emad Abbas Jaffar Al-Mulla, Fayq Hsan Jabbar, Zaidoon Jawad Kadhim, Ali Abdulabbas Abdullah, Ahmed Ghanim Wadday, and Sabah Mohammed Mlkat. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.