Article
The Three-Dimensional Images and Intracellular Calcium Analysis of Weigela Floridacv and Lonicera Japonica Thunb Pollen
Dongwu Liu 1, 2*, Zhiwei Chen 1, 2, Xue Wang 2, Hongzhi Xu 1, Lina Wang 1
1Analysis and Testing Center, Shandong University of Technology, Zibo, Shandong Province 255049.
2School of Life Sciences, Shandong University of Technology, Zibo, Shandong Province 255049.
*Corresponding authors. Email: liudongwu@sdut.edu.cn
Citation: D. Liu, et al., The Three-Dimensional Images and Intracellular Calcium Analysis of Weigela Floridacv and Lonicera Japonica Thunb Pollen. Nano Biomed. Eng., 2009, 1(1): 57-60.
DOI: 10.5101/nbe.v1i1.p57-60
Abstract
Confocal microscope, which is a major advance upon normal light microscope, has been used in a number of scientific fields. Moreover, the three dimensional images of specimens can be reconstructed with confocal microscope. It is ideal to analyze the three dimensional specimens for the nondestructive, noninvasive nature of the confocal microscope. In the present studies, a series of Weigela floridacv and Lonicera japonica thunb pollen optical sections were acquired with confocal microscope. Then the three-dimensional images of the pollen were reconstructed with the software of confocal microscope. In addition, intracellular calcium in the pollens was detected with the probe Fluo-3 AM, and the distribution of calcium in the pollens was analyzed with confocal microscope. Our results indicate that it is a very easy job to analyze the three dimensional digital images of the pollen and intracellular calcium in the pollens with confocal microscope and the probes Acridine orange (AO) and Fluo-3 AM.
Keywords: Confocal microscope; Pollen; Three-dimensional image; Reconstruction; Calcium
1. Introduction
Confocal microscope, which is one of the most exciting advances in optical microscope, has become a routine technique and indispensable tool for cell bio- logical studies and molecular investigations [1]. Compared with conventional microscope, the specimens can be analyzed in three dimensions with much more clarity with confocal microscope. In addition, thick and opaque specimens that can not be observed in a conventional light microscope are excellent specimens for a confocal microscope. Confocal microscope is ideal to analyze the three dimensional specimens because its non-destructive, non-invasive nature allows imaging of a sample at a sequence of depths without physical sectioning. The potential roles of Ca2+ in pollen have received considerable attention in the recent years. The signals of Ca2+ are thought to play an important role in plant growth and development, including key aspects of pollen tube growth and fertilization [2]. It has been reported that pollen germination and pollen tube growth has significantly regulated by the transport of Ca2+ [3-4]. Confocal laser scanning microscope has been widely used to study calcium in the pollen tubes [5]. However, there are fewer reports about the distribution of Ca2+ in pollens. There have been numerous scientific researches employing confocal microscope in the pollen studies, such as magnitude and direction of vesicle dynamics in the growing pollen tubes, mitochondrial morphology in pollen, and the actin cytoskeleton of the pollen [6-8]. Since the primary value of the confocal microscope is its ability to produce optical sections through a 3-dimensional specimen, the aims of the present studies gela floridacv and Lonicera japonica thunb pollens. Furthermore, the distribution of Ca2+ in the pollens was investigated in the present studies.
2. Materials and Methods
2.1 Plant material and reagents
Pollen was collected from the Weigela floridacv and Lonicera japonica thunb plants grown in Shandong University of Technology. The pollens were placed in a 5.0 mL micro-centrifuge tube and 4.0 mL of 20 μM Acridine orange (AO) (Molecular Probes, Inc) was added. The micro-centrifuge tube was incubated in dark at 4C for 1 h followed by incubation at 20C for 1 h in dye free solution. To analyze Ca2+ level, pollens were loaded with 5 μM Fluo-3 AM (Molecular Probes, Inc) at 37C for 30 min. Excess dye was eliminated by washing the disks several times in phosphate buffer.
2.2 Laser scanning confocal microscopy
To reconstruct the three dimensional images, the pollen was observed with a 40× lens and the images were captured with a confocal microscope. A laser scanning confocal microscope (Leica TCS SP2, Germany) with an air cooled, argonion laser as the excitation source at 488 nm was used to view the pollen. The series images of pollen were detected in the yellow channel. In addition, the excitation source at 488 nm was used to view the sites of Ca2+, and the distribution of Ca2+ were detected in the green channel. The channel settings of pinhole, detector gain, amplification offset and gain were adjusted to provide an optimal balance of fluorescence intensity of the targeted pollen and background. Data were collected by a computer attached to the instrument, stored on the hard drive, processed with a Leica TCS Image Browser, and transferred to Adobe Photoshop 6.0 for preparation of Figure.
2.3 Three dimensional pollen images reconstruction
Sixty-three Weigela floridacv pollen optical sections and fifteen Lonicera japonica thunb pollen optical sections were acquired with the laser-scanning confocal microscope, respectively. Then the software of Leica TCS SP2 was used to reconstruct the three-dimensional images of the Weigela floridacv and Lonicera japonica thunb pollen.
3. Results and Discussion
Confocal microscope is a relatively new light microscopical imaging technique, which has been widely used in the biological sciences. One of the important functions to the biologist is its ability to produce opti. Furthermore, the three-dimensional images are accurate representations of the data and can be handled and manipulated to evaluate surface details and morphology. There is a pinhole aperture in the confocal microscope, which is confocal with the specimen point. The pinhole blocks light from other points in the specimen and permits the imaging of a well-defined spot deep inside the specimen. With moving mirrors and motion of the objective, a three-dimensional image can be constructed by scanning this spot [9]. Then a series of optical sections can be transformed into a three-dimensional volume. The volume permits visualization of the stack from viewpoints, which are different from the plane containing the individual optical sections [10]. In addition, the reconstruction of three-dimensional image by optical sectioning is faster and more precise than the mechanical sectioning. Another thing, the interior of relatively thick histological sections can be analyzed with confocal microscope [11-15]. Thus the morphology and differentiation of thicker sections can be provided [14, 15]. In the present studies, sixty three Weigela floridacv pollen optical sections and fifteen Lonicera japonica thunb pollen optical sections were acquired with the laser scanning confocal microscope, respectively (Figure 1 and 2). Then the three dimensional images of the Weigela floridacv and Lonicera japonica thumb pollen were reconstructed with confocal microscope software (Figure 3, A and B). Here we applied AO to dye the pollens of Weigela floridacv and Lonicera japonica thunb, and the clear 3-dimensional image of the pollen was acquired (Figure 3, A and B). Our results indicate that it’s a very easy job to analysis pollen with confocal microscope and the probe AO. Confocal microscopy provides the means to localize molecules in living cells with high spatial and temporal resolution. Since calcium is an intracellular messenger, the techniques for measuring cytosolic free Ca2+ concentrations have been essential. Fluo-3 fluorescence depends on concentration of free Ca2+ [16, 17]. Using Fluo-3 AM as probes, the distribution of Ca2+ can be clearly detected in the pollens of Weigela floridacv and Lonicera japonica thunb (Figure 4, A and B). Moreover, the results indicated that there is more Ca2+ distri- bution in the periphery of Weigela floridacv and Lonicera japonica thunb pollens (Figure 4, A and B). Taken together, our results indicate that it is a good method to research the pollens with confocal microscope. With the fluorescence probes AO and Fluo-3 AM, not only the 3-dimensional image of the pollen can be reconstructed, but also the fluorescence of Ca2+ can be analyzed. It’s a very easy job to analyze three-dimensional images of the pollen and intracellular calcium in the pollens with confocal microscope and the probes AO and Fluo-3 AM.
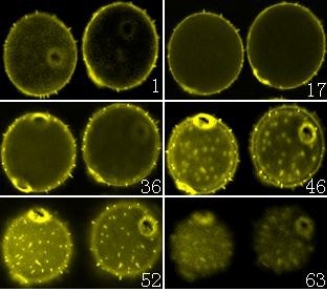
Figure 1. A series of confocal microscope optical sections of Weigela floridacv pollen. Pollen was labeled with AO. Sixty three optical sections were acquired with the laser-scanning confocal microscope, and six images were chosen in the Figures (from No.1 to No.63 image). (×400).
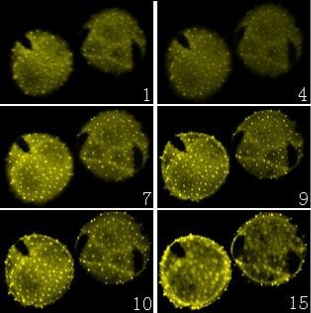
Figure 2. A series of confocal microscope optical sections of Lonicera japonica thunb pollen. Pollen was labeled with AO. Fifteen optical sections were acquired with the laser-scanning confocal microscope, and six images were chosen in the Figures (from No.1 to No.15 image). (×400).


Figure 3. The three-dimensional images of the pollen were reconstructed with confocal microscope software.(A) The three dimensional images of the Weigela flor- idacv pollen. (B) The three-dimensional images of the Lonicera japonica thunb pollen. (×400).

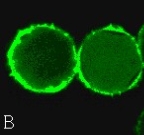
Figure 4. The distribution of calcium in the pollens. (A) The distribution of calcium in the Weigela floridacv pollens. (B) The distribution of calcium in the the Lonicera japonica thunb pollens. (×400)
Acknowledgements
This work was supported by the Foundation for technological innovation of experimental instruments and equipment in Shandong Province (Grant No. 2008GG2TC01011-5) and the National Natural Science Foundation of China (Grant No.30900071).
References
1. Lichtman JW. Confocal microscopy. Scientific American 1994; 271: 40–53. doi:10.1038/scientificamerican0894-40
2. Hepler PK, Vidali L, Cheung AY. Polarized cell growth in higher plants. Annu Rev Cell Dev Biol 2001; 17: 159–187. doi:10.1146/annurev.cellbio.17.1.159
3. Yamaoka S, Leaver CJ. EMB2473/MIRO1, an Arabidopsis Miro GTPase, is required for embryogenesis and influences mitochondrial morphology in pollen. Plant Cell 2008; 20: 589–601. doi:10.1105/tpc.107.055756
4. Liu ZQ, Xu GH, Zhang SL. Pyrus pyrifolia stylar S-RNase induces alterations in the actin cytoskeleton in self pollen and tubes in vitro. Protoplasma 2007; 232: 61–67. doi:10.1007/s00709-007-0269-4
5. Roy SJ, Holdaway Clarke TL, Hackett GR, Kunkel JG, Lord EM, Hepler PK. Uncoupling secretion and tip growth in lily pollen tubes: evidence for the role of calcium in ex- ocytosis. Plant J, 1999, 19: 379–386. doi:10.1046/j.1365-313X.1999.0 0515.x
6. Feijó JA, Malho' R, Obermeyer G. Ion dynamics and its possible role during in vitro pollen germination and tube growth. Protoplasma 1995; 187: 155–167. doi:10.1007/BF01280244
7. Tayler LP, Hepler PK. Pollen germination and tube growth. Annu Rev Plant Physiol Plant Mol Biol 1997; 48: 461–491.
doi:10.1146/annurev.arplant.48.1.461
8. Bove J, Vaillancourt B, Kroeger J, Hepler PK, Wiseman PW, Geitmann A. Magnitude and direction of vesicle dy- namics in growing pollen tubes using spatiotemporal image correlation spectroscopy and fluorescence recovery after photobleaching. Plant Physiol 2008; 147: 1646–1658. doi:10.1104/pp.108.120212
9. Dinsmore AD, Weeks ER, Prasad V, Levitt AC, Weitz DA. Three-Dimensional Confocal Microscopy of Colloids. Ap- plied optics 2001; 40: 4152–4159. doi:10.1364/AO.40.0 04152
10. Van der Voort HT, Brankenhoff GJ, Baarlslag MW. Three-dimensional visualization methods for confocal mi- croscopy. J Microsc 1989; 153: 123–132.
11. McCauley MM, Hepler PK. Cortical ultrastructure of freeze-substituted protonemata of the moss Funaria hygro- metrica. Protoplasma 1992; 169: 215–230. doi:10.1007/BF 01323616
12. Gray JD, Kolesik P, Hoj PB, Coombe BG. Confocal meas- urement of the three-dimensional size and shape of plant parenchyma cells in a developing fruit tissue. Plant J 1999; 19: 229–236. doi:10.1046/j.1365-313X.1999.0 0512.x
13. Funada R, Abe H, Furusawa O, Imaizumi H, Fukazawa K and Ohtani J. The orientation and localization of cortical microtubules in differentiating conifer tracheids during cell expansion. Plant Cell Physiol 1997; 38: 210–212.
14. Kitin P, Funada R, Sano Y, Ohtani J. Analysis by Confocal Microscopy of the Structure of Cambium in the Hardwood Kalopanax pictus. Ann Bot 2000; 86: 1109–1117. doi:10.10 06/anbo.2000.1281
15. Kitin P, Sano Y, Funada R. Fusiform cells in the cambium of Kalopanax pictus are exclusively mononucleate. J Exp Bot 2002; 53: 483–488. doi:10.1093/jexbot/53.368.483
16. Kao JP, Harootunian AT, Tsien RY. Photochemically gen- erated cytosolic calcium pulses and their detection by fluo-3. J Biol Chem 1989; 264: 8179–8184.
17. Minta A, Kao JP, Tsien RY. Fluorescent indicators for cytosolic calcium based on rhodamine and fluorescein chromophores. J Biol Chem 1989; 264: 8171–8178.
Received 10 November, 2009; accepted 6 December, 2009; published online 9 December, 2009.
Copyright: (c) 2009 D. Liu et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.